

PLASMA ETCHING OF HfO2 GATE-STACKS WITH Si SELECTIVITY*
Juline Shoeb** and Mark J. Kushner***
**Department of Electrical and Computer Engineering
Iowa State University, Ames, IA 50010
,
***Department of Electrical Engineering and Computer Science
University of Michigan, Ann Arbor, MI 48109
1. HfO2 Gate-Stack
In this project, results from integrated reactor and feature scale modeling of gate stack etching in Ar/BCl3/Cl2 plasmas, preceded by photo-resist trimming in Ar/O2 plasmas, are discussed. It was found that BCly species produced by electron impact in the plasma react with HfO2, which, under ion impact, form volatile etch products such as ByOClx and HfCln. Selectivity to Si is achieved by creating Si-B bonding as a precursor to the deposition of BCly polymer, which slows the etch rate relative to HfO2. Validation was performed with data from the literature. The effect of bias voltage and key reactant probabilities on etch rate, selectivity and profile were also investigated. The Hybrid Plasma Equipment Model (HPEM) was used to predict the reactor scale plasma characteristics and reactant fluxes to surfaces in the ICP reactor. The Monte Carlo Feature Profile Model (MCFPM) was used to address plasma surface interactions and evolution of surface morphology and profiles of the gate-stack. The initial gate-stack used for the work is shown in Fig. 1.
 |
| Fig. 1: Initial gate-stack consisting of PR, BARC, TiN, HfO2, SiO2 and Si substrate. |
2.Reaction Mechanism
Fundamental principles such as bond energies and volatility of the etch products were considered to develop the HfO2 etch mechanism in Ar/Cl2/BCl3 plasmas. HfO2 etching is followed by the breaking of two Hf-O bonds, each having bond strength of approximately 8.3 eV. Ions having sufficient energy break one Hf-O bond, and hence generate separated HfO (s) and O(s), where (s) denotes a surface species. Ions having higher ion energy than the threshold can break the remaining Hf-O bond and thus can separate O(s) and Hf(s). Hf(s) later adsorbs Cl to produce HfCl(s), HfCl2(s), HfCl3(s) and HfCl4(s). HfCln(s) species are removed as volatile HfCln(g) (n=2,3,4) species by ion impact, where (g) denotes a gas phase species. Key reactions for HfO2 etching can be summarized as
M+(g) + HfO2(s) > HfO(s) + O(s) + M(g), p= 0.4, Et = 14 eV M+(g) + HfO(s) > Hf(s) + O(s) + M(g), p = 0.4, Et = 14 eV Cl(g) + Hf(s) > HfCl(s), p = 0.2 M+(g) + HfCln(s) > HfCln(g) + M (g), p = 03-0.9, Et = 14eV where p denotes the probability of the reaction and Et is the threshold energy of the reaction and M+(g) is an ion. Formation of Si-B bonds enable the deposition of BCly(g) species as polymer on the top of Si-B bonds and thereby ensure high selectivity. However, the ion energies striking the surface should be sufficiently low to avoid sputtering of the passivation layer. A summary of the reactions for Si-B bond formation and polymer deposition are BCly (g) + Si (s) > SiBCly (s), p = 0.4 BCly (g) + SiClz (s) > SiClzBCly (s), p = 0.4 BCly (g) + SiBCly (s) > SiBCly(s) + Polymer(s), p = 0.7 BCly (g) + SiClzBCly (s) > SiClzBCly(s) + Polymer(s), p = 0.7 TiN(s) etching is analogous to other metals with volatile etch products. Energetic ions break the bonding between Ti and N and so enable Ti(s) to adsorb neutral Cl(g). Such adsorptions lead to the formation of TiClx(s) (x=1, 2, 3). Later energetic ions can remove TiCln(s) as volatile TiCln(g) (n=2,3,4). M+(g) + TiN(s) > Ti(s) + N(g) + M(g), p=0.51, Et = 10 eV M+(g) + TiCln(s) > TiCln(g) + M(g), p = 0.35-0.65, Et = 10 eV3. Results
The effect of bias voltage on HfO2 etch rate was investigated. As expected, the etch rate is approximately proportional to the square root of bias voltage. The post-etch gate-stack profiles, etched under three different bias voltages are shown in Fig. 2.
 |
| Fig. 2: HfO2 etch rate and selectivity as a function of bias voltage. HfO2/Si selectivity degrades with increase in bias voltage. |
The importance of Si-B bond formation that later initiates the deposition of polymer was investigated. Si-B bonds are formed due to the adsorption of neutral BCly(g) by Si(s) and SiClz(s) (z =1, 2, 3) species. Three gate stacks etched with a 32 V bias with different Si-B bond formation probabilities are shown in Fig. 3. When the probability of this adsorption reaction is 0, the selectivity is approximately 1. As this probability increases from 0 to 0.4, etching of Si slows down and the selectivity improves to 13. However, further increase in this probability does not increase the selectivity. This specific test validates the importance of Si-B bonds for obtaining a good selectivity.
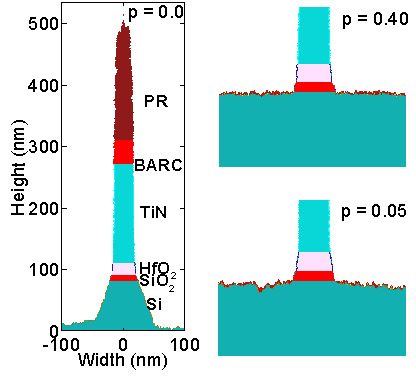 |
| Fig. 3: Si-B bond formation effect shown for gate-stacks etched with different Si-B bond formation probabilities. Selectivity improves as probability increases. |
To demonstrate the effect of polymer formation on selectivity between Si and HfO2, the polymer deposition probability was also varied from 0 to 1.0 with a 32 V bias. As the probability is increased from 0 to 0.5, the selectivity improves from 3 to 10 and is almost constant at probabilities higher than 0.5. Post-etch profiles of the gate-stack etched with three different polymer formation probabilities are shown in Fig. 4.
 |
| Fig. 4: Polymer formation effect on HfO2/Si selectivity shown for three gate-stacks etched with different Si-B bond formation probabilities. Selectivity improves as probability increases. |
*Work supported by Semiconductor Research Corp.
Last updated: March 01, 2009.