I. Conventional Oxygen-Iodine Lasers
Chemical oxygen-iodine laser (COIL) operation on the I(2
P1/2) » I(2P3/2)
electronic transition in iodine has been pursued for more than three decades because of its high efficiency
and potential multi-kilowatt cw power. The relatively short 1.315 micron wavelength is especially
attractive because of its high optical coupling with most common metals and high transmission in optical
fiber. These properties are desirable for such uses as anti-missile defense systems and remote
dismantling of nuclear reactors. The traditional chemical oxygen-iodine laser (COIL) achieves
I2 dissociation and ionization in a supersonic nozzle via I2 injection into a stream
of excited singlet-delta O2. Some of the excited O2 dissociates the molecular
iodine; the rest then excites the atomic iodine in a near resonant transfer to create population inversion
and lasing on the I(2P1/2) »
I(2P3/2) transition.
II. ElectriCOIL
Conventional COILs generate the singlet-delta O2 metastable at yields up to 70% by reaction
of Cl2 in basic H2O2. However, this method is less attractive for
airborne missile defense systems because of the complexity, high weight, and operational hazards associated
with the liquid chemical storage and pumping systems. For airborne systems, low system weight demands
and reduced complexity have driven the development of all gas phase singlet-delta O2 generators.
Research has shown that singlet-delta O2 yields up to 32% can be produced by rf
discharges along with significant quantities of atomic oxygen, singlet-sigma O2, and
O3. This type of electrical discharge COIL has been termed the ElectriCOIL
(Fig. 1).
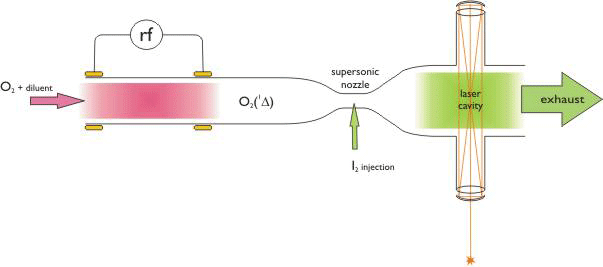 |
| Figure
1. Oxygen-iodine laser with electric discharge singlet-delta O2
production. |
Despite the reports of yields in 20 - 30% range, the singlet-delta O2 has not been
produced at high enough pressures and flow rates at sufficiently low temperatures to insure population
inversion in iodine. Thus, no ElectriCOIL experiment has yet demonstrated laser gain, and more
research is needed to understand the oxygen discharge electrodynamics. At UIUC we are simulating
gas phase singlet-delta O2 generation via rf discharges in pure O2 and in
mixtures with inert diluents such as He.
III. GlobalKin Model
To model this complex reactive system we are using GlobalKin, a software package developed at UIUC
for zero-dimensional batch plasma reaction kinetics that has been modified to simulate temporally
invariant one-dimensional plug flow. Using a specified species and reaction set, GlobalKin
calculates reaction rates for various neutrals and ions and integrates them along the plug flow
length. In addition, various electron impact reaction rates are calculated for specified power
depositions in the discharge region, where GlobalKin first solves the spherically symmetric Boltzmann
equations for the electron energy distributions (Fig. 2).
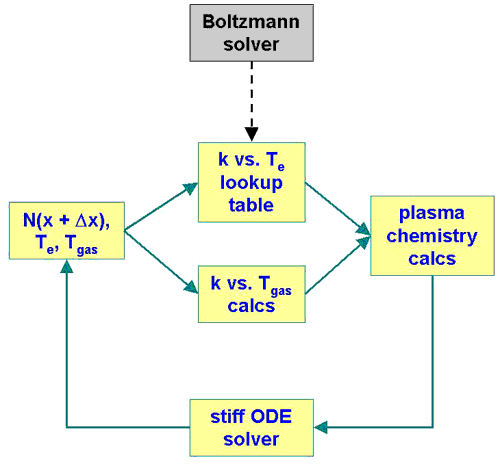 |
| Figure 2. GlobalKin
program flow. |
The GlobalKin results are validated with experiments on an ElectriCOIL run by
CU Aerospace and the
UIUC Chemical Laser Group.
Various parameterizations are run based on discharge length, power deposition, system pressure,
diluent ratios, etc. which are then used to direct further experimental work on the ElectriCOIL.
Fig. 3 shows some recent results from the simulation of a 20 cm long discharge
in pure O2.
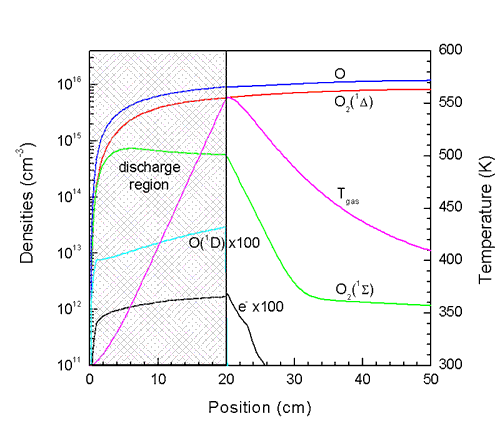 |
| Figure 3. Species
densities and gas temperature in a 20 cm discharge in pure O2
at 3 Torr. Inlet velocity is 1000 cm/s and power deposition is 0.5
W/cm3. |
IV. Results
Results of a large scale parameterization have verified that singlet-delta O2 yield
scales primarily with specific energy deposition in the gas, where the specific energy is taken on
an inlet O2 basis. Yield increases linearly at low specific energy deposition,
rising to a maximum at 5-8 eV/O2 molecule before falling off again as dissociation into
O atoms dominates. Fig. 4 shows a small range of the singlet-delta
O2 and atomic O yield results from the parameterization. Actual specific energy
depositions ranged to 250 eV/O2 molecule, continuing the trends shown in
Fig. 4.
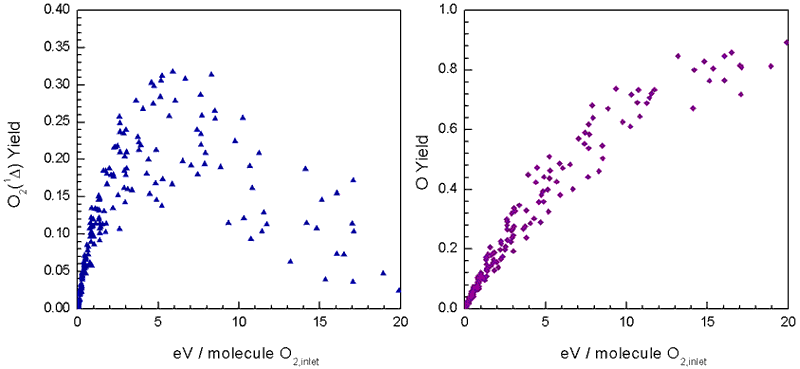 |
| Figure 4. Singlet-delta
O2 and atomic O yield results from a 256 run parameterization
on discharge velocity, pressure, power, and diluent (He) ratio. |
Although the singlet-delta O2 yield is clearly a strong
function of specific energy deposition, there is still a large amount
of variance between runs at identical specific energies. This
remaining scatter can be attributed to diluent, pressure, and power
effects. The combined effects of O2 partial pressure
and mixture ratio on singlet-delta O2 yield are shown in Fig. 5. The animation shows how the yield
increases as the partial pressure of O2 increases, but in
all cases the peak yield occurs when large amounts of the He diluent
are present. This can be attributed to the lower reduced electric
field (E/N) caused by increasing pressure and by addition of diluent,
allowing the discharge to operate at a more efficient E/N for
excitation of singlet-delta O2.
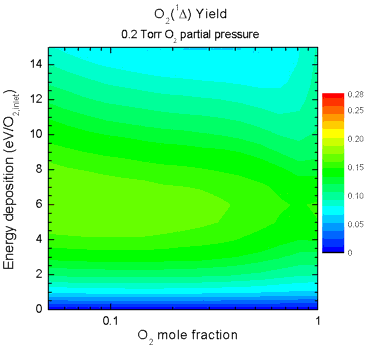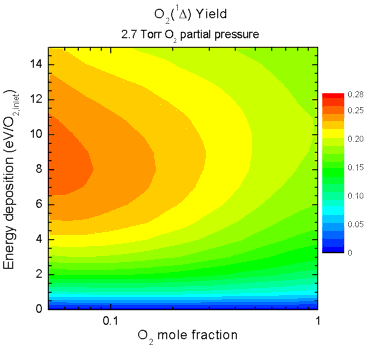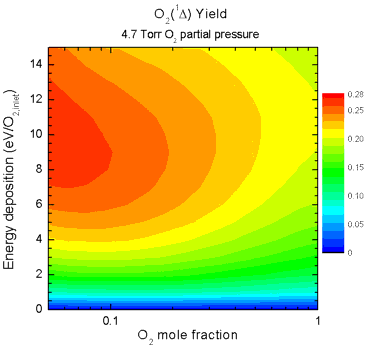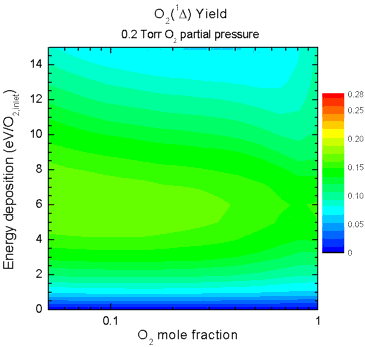 |
| Figure 5. Animation
showing yields as a function of O2 mole fraction (abscissa)
and specific energy deposition (ordinate) as the O2 partial
pressure is increased from 0.2 - 7.2 Torr. |
Reducing the specific power deposition of the discharge also increases
singlet-delta O2 yield, as shown in Fig.
6. The yield shows a peak near 1 W/cm3, suggesting
that very low powered discharges are not necessary to achieve the best
yields, and indeed may not be capable of sustaining the glow.
Power depositions of 1 W/cm3 (0.33 W/cm3/Torr O2
in Fig. 6) are still low compared to current
ElectriCOIL technology, and would require moderately long discharges
(~1 m) to achieve the 5 - 8 eV/molecule O2 necessary to
obtain the best yield.
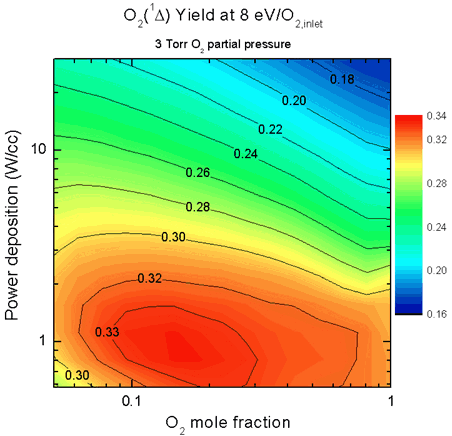 |
| Figure 6. Contour
plot showing the peak in singlet-delta O2 yield at low specific
power deposition. |
Although the GlobalKin model reproduces experimental results
adequately, it is still inherently a zero-dimensional model.
Currently, a full one-dimensional time variant model is in
development. Future research will more accurately describe the
effect of specific energy deposition as well as the secondary effects
of diluents, pressure, and power deposition using the 1-D model.
1This work is supported by the
National Science Foundation
(CTS 99-74962, CTS03-15353) and
AFOSR/AFRL.
Updated 8.27.2003






